Product
제품
-
Analytical Products
- Wyatt Technology
- Awareness Technology
- Eurofins l Abraxis (GSD)
- Aurora Biomed
- Canada NRC-CNRC
- Cifga
- Chrom Tech
- Eichrom Technologies
- EPROGEN
- Fluidic Analytics
- Fianovis
- Globe Scientific
- GENERON
- Hygiena International
- Halo Labs
- KROMATON
- InProcess-LSP
- MTC Bio
- MZ-Analysentechnik
- Newomics Inc.
- Occhio Instruments
- Optimize Technologies
- Pickering Laboratories
- PolyLC
- Raykol Group
- RheoSense
- Rocker Scientific
- Santai Science
- SEDERE
- Spectra Analysis
- Spectradyne
- UCT
- Wealtec Corp
-
Bio & Medical Products
- Biolog
- Adooq Bioscience
- A&A Biotechnology
- Accegen Biotechnology
- Anatrace
- Array Bridge
- Biogenes GmbH
- BioQuochem
- BioServ UK
- Biomiga
- Biotech Support Group
- CinderBio
- Cell Technology
- Creative Biolabs
- Creative Diagnostics
- Creative Biostructure
- Creative Biomart
- Creative Enzymes
- EICOM
- Emulseo
- GLYcoDiag
- Helix Biotech
- InnoGenomics
- IsoSciences, LLC
- IUL Instruments
- Micropore Technologies
- Matrix Innovation
- PreciGenome
- PhylumTech
- ProFoldin
- Protein Ark
- Primer Design
- ProteoChem
- RareCyte
- RECIPE
- Silicycle Inc.
- Tymora Analytical
- UTAK
- YouSeq
- Z Biotech
- AlbuVoid™ PLUS
- 제품명: AlbuVoid™ PLUS
- 용도: Albumin & IgG Removal for Proteomics
- 메이커: Biotech Support Group
- 카달로그:
소개
AlbuVoid™ PLUS Albumin and IgG Depletion From Serum/Plasma for Proteomics
• IgG removal >90% (70-80% of total Immunoglobulins removed)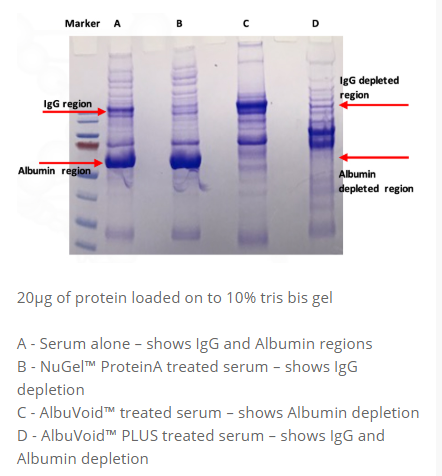
• Albumin removal >95%
• Seamless and simple < 1 hour protocol
• Low abundance enrichment equivalent to immuno-affinity
• Disposable, cost-effective, no column regeneration or cross-contamination
• Works for most species tested including human, sheep, rat, mouse, bovine
• On-bead protocols improve workflow and efficiency, especially suited to
targeted proteomics
• Suitable for LC-MS, 1 and 2D Gels, ELISAs, Enzyme and other Functional
Assays.
The classical plasma proteins generally fall into functional categories, forming
the vast majority of the mid-to-high abundance proteome. In serum, these
sub-proteomes by mass content are: Albumin 50-60%; Immunoglobulins 10-20%;
Transport Proteins (Transferrin, Apo) 5-10%; Complement related Proteins 3-5%;
Protease Inhibitors 2-5%; and all others 2-5%. While these sub-proteomes are
required for normal body homeostasis, they nevertheless can become dysfunctional
during acute-phase and chronic stimuli.
So, depending on the needs of the investigation, it can be valuable to consider that one or more of these categorical sub-proteomes is simply background noise whereby depletion is beneficial. While in other cases, these same categorical sub-proteomes might provide new data and information and consequently, should not be depleted. Different AlbuVoid™, AlbuVoid™ PLUS and AlbuSorb™ PLUS workflows support different proteomic biases as outlined in the following Table.
Products and digest conditions produce different sub-proteome windows of observation. So, depending on the needs of the investigation, it can be valuable to consider that one or more of these categorical sub-proteomes is simply background noise whereby depletion is beneficial. While in other cases, these same categorical sub-proteomes might provide new data and information and consequently, should not be depleted. Categorically the acute-phase sub-proteomes differentiated in disease may vary greatly from those associated with chronic sub-proteomes. So there is great benefit in having options to enrich or deplete one or more of these sub-proteomes.
BSG's Albumin and IgG Removal Kits offer many such options:
• The 'PLUS' products substantially deplete Immunoglobulins through separations at the protein level.
• The variable regions of Immunoglobulins are extremely heterogeneous, generating a background noise across the full LC gradient.
On-bead digestion (BASP™) with AlbuVoid™ substantially reduces the influence of such Ig peptide features. So in addition to
workflow simplicity, BASP™ can be advantageous utilized in targeted proteomic workflows whenever the target proteins do not
require strong denaturing conditions.
• With the exception of Immunoglobulins whereby FASP generates many more spectral features, both strong denaturing conditions
(FASP) and on-bead digest (BASP™) conditions produce similar protein profiles.
• Both Apolipoproteins and heavily glycosylated proteins (i.e., α1-Acid Glycoprotein) bind poorly to AlbuVoid™.
For quantitative studies within these classes of proteins, AlbuSorb™ PLUS is recommended.
• The Complement sub-proteome is especially enriched by AlbuVoid™ PLUS. The digest conditions may bias towards one or more
functional sub-populations, likely due to conformational transitions and protein-protein interactions (i.e., Factor Bb, Properdin)
that occur upon activation. This needs further investigation.
• The low abundance sub-proteome is enriched 5+ fold with AlbuVoid™ and 4+ fold with AlbuSorb™ PLUS.
☞ Click Here For The AlbuVoid™ Plus Product Sheet.

사양
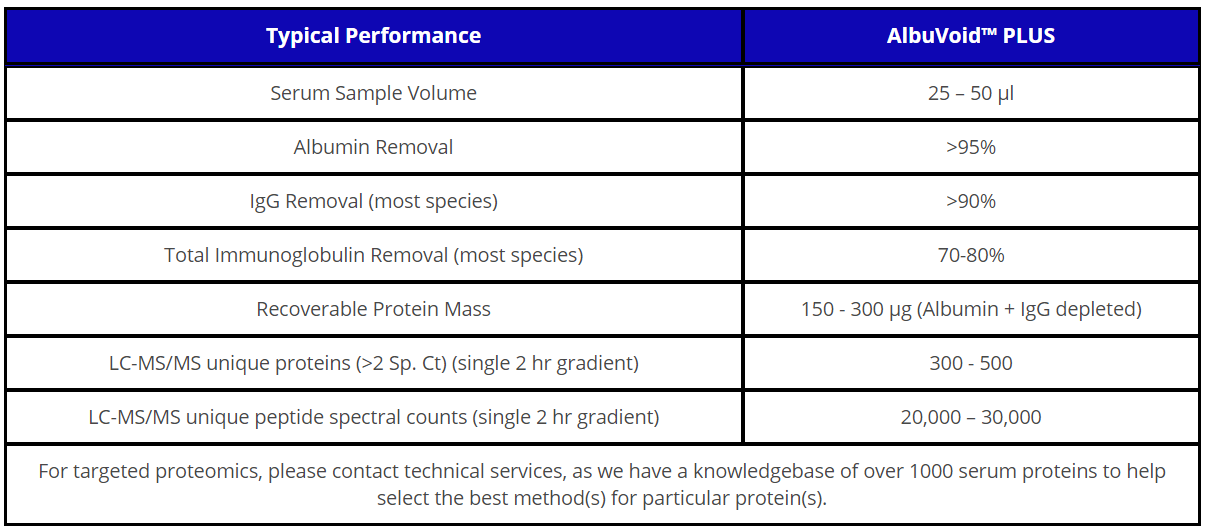
주문정보

Kit Components :
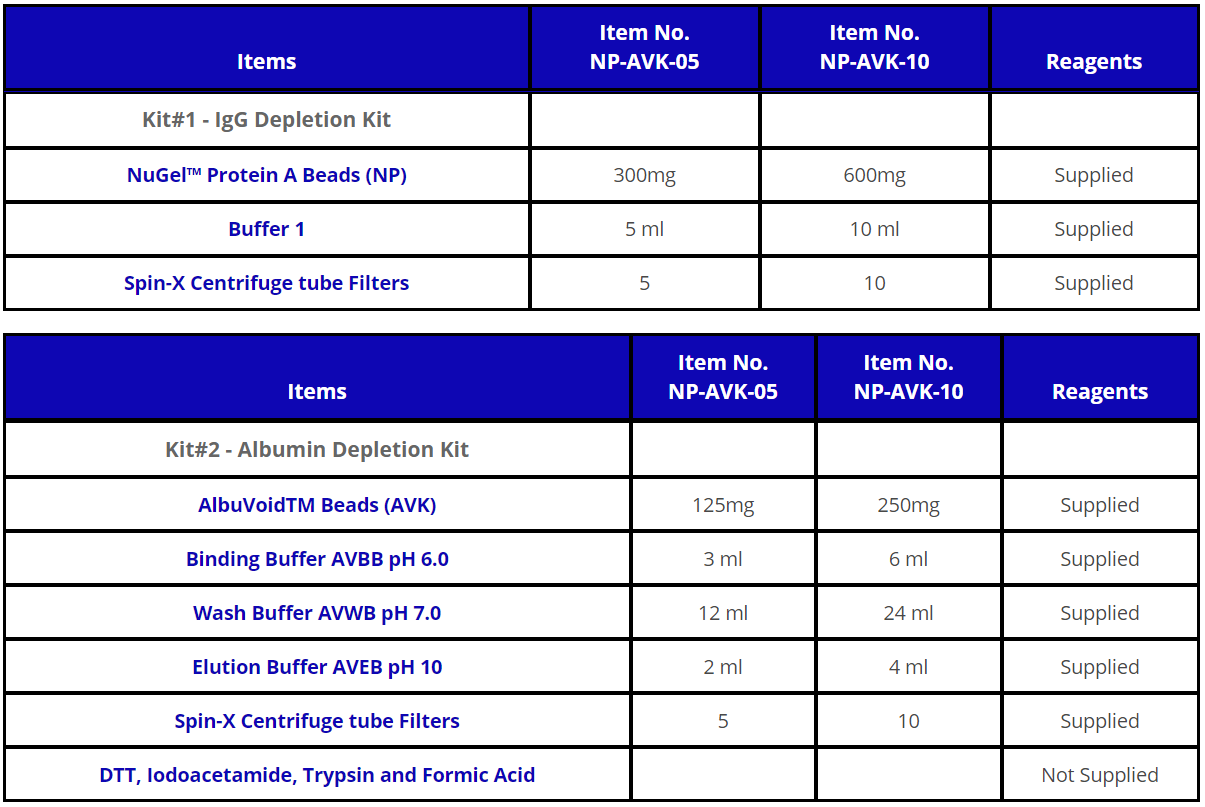
관련자료
References
AlbuVoid™ PLUS & AlbuSorb™ PLUS - Evaluating Different Windows of Observation Solves The Many Challenges of Serum Proteomics
Zheng H, Soherwardy A, Roy S, Kuruc M, Avadhani S. AlbuVoid™ Enrichment & Antibody Depletion – Tackling the Challenges of Serum Proteomics Part II. Poster reprint first presented at 26th International Molecular Med TRI-CON, March 10-15, 2019, San Francisco, CA USA
Vialaret, Jerome & Kadi, Sarah & Tiers, Laurent & O Flynn, Robin & Lehmann, Sylvain & Hirtz, Christophe. (2018). Albumin depletion of human serum to improve quantitative clinical proteomics. Current Topics in Peptide & Protein Research 19. 53-62.
Poillet-Perez, Laura, et al. "Autophagy maintains tumour growth through circulating arginine." Nature (2018): 1.
Roy, Swapan, and Matthew Kuruc. "Methods to Monitor the Functional Subproteomes of SERPIN Protease Inhibitors." Functional Proteomics. Humana Press, New York, NY, 2019. 41-54.
Mihara, Keisuke, et al. "Identification of Specific Protein Markers of Rheumatoid Arthritis in Synovial Fluid and Serum." Journal of Hard Tissue Biology 27.1 (2018): 55-58.
David L. Wang, Chuanguang Xiao, Guofeng Fu, Xing Wang and Liang Li. Identification of potential serum biomarkers for breast cancer using a functional proteomics technology. Biomarker Research (2017) 5:11. DOI 10.1186/s40364-017-0092-9
Zheng, H., et al. "AlbuVoid™ coupled to on-bead digestion-tackling the challenges of serum proteomics." J Proteom Bioinformatics 8.9 (2015): 225.
Zheng, H., et al. Poster reprint first presented at 14th Human Proteome Organization World Congress (HUPO 2015), held on September 27 – 30, 2015 entitled “New Proteomic Workflows Combine Albumin Depletion and On Bead Digestion, for Quantitative Cancer Serum”.
